Our brands
From health to beauty
From health to beauty

Good health is a state of complete physical and mental well-being, which cannot be reduced to the simple absence of illness. Pierre Fabre's philosophy, forged throughout his career as a pharmacist, was always to consider health and beauty as forming an indissociable whole, demanding the same ethical and scientific approach.
On this matter, Pierre Fabre, when a pharmacist in the Tarn area of south-west France, said “We strive for health, life, beauty - this is where the
nobility of our profession lies. And in my opinion, it is one of the most exciting professions. However, we must do this with great modesty and a moral sense in keeping with the ethics of pharmacy. It is a great thing to develop a prescription drug to treat patients or relieve their pain, it is gratifying
to be able to contribute in a serious manner to enhancing the beauty of a face or a body” 50 years later, the challenge has been met.
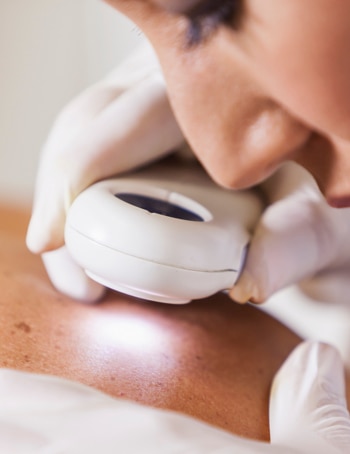
Caring for
cancer
Caring for
cancer
From the launch of a chemotherapy product derived from the Madagascar periwinkle in the late 1980s, to the development of an “oral” form of this treatment to allow it to be administered at the patient’s home, and more recently, the development of a new targeted therapy for patients suffering from advanced melanoma... the Pierre Fabre Group makes oncology a major priority.

Dermo-cosmetics
More than 50 years ago, Pierre Fabre invented dermo-cosmetics, a pioneering vision that brings together health and beauty.
Ever since, we have been working daily to offer you the most suitable and most innovative solutions to take care of your hair and skin, in order to improve their dermatological condition with lasting effect and to reveal their beauty.
We respect nature, the source of inspiration for our botanists and agronomists.
We constantly innovate alongside health professionals.
We test every formula with the utmost stringency, to ensure your safety.
We manufacture our products in France.
Because looking after our skin and hair, using products that are in harmony with our own nature and nature in general, is a first step toward overall better-being. This is what we are all about.

Caring for the skin
Caring for the skin
Because beautiful skin is above all healthy skin, our products provide the solution adapted to your needs.

iconic brands for beautiful, healthy skin

Caring for hair
Caring for hair
Shine, bounce, strength, density, eliminating dandruff and addressing hair loss... Having the hair of your dreams involves a whole host of aspects. We have gone through them with a fine-tooth comb to ensure that our care products are effective for you!