Pharma R&D
A patient-driven integrated research
A patient-driven integrated research
We design and develop innovative solutions in collaboration with partners and health care professionals, constantly drawing our inspiration from nature and plants, always placing the patient at the heart of our research and development.
Pharmaceutical R&D figures
Pharmaceutical R&D figures
Medical and scientific innovation center at the Oncopole in Toulouse
people dedicated to Pharma R&D
dedicated to Pharma R&D in 2025
Scientific Advisory Board
The Scientific Advisory Board is composed of internationally recognized experts. It meets periodically to assess the research and development strategy of new therapeutic assets and partnerships in medical innovation.
Our pipeline
Pierre Fabre Group researchers work tirelessly to develop innovative treatments and technologies in the fields of oncology and dermatology.
Discover all our molecules currently in development.
Download the file
Thank you for the interest you give to our publications. Before accessing to our document, please tell us more about you.
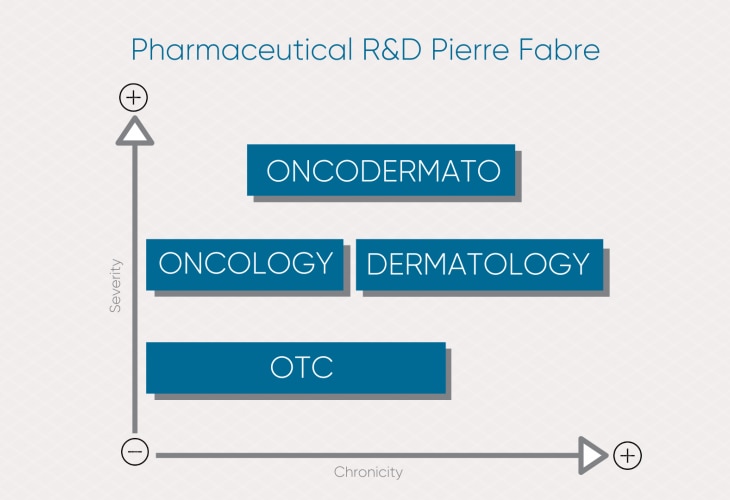
Pharmaceutical research areas
Pierre Fabre invests in 4 lines of pharmaceutical research to devise innovative solutions and provide therapeutic solutions for unmet needs.
Our integrated, cross-disciplinary research, from health to beauty, generates value for patients and health care professionals.
Oncology R&D
Cancer is currently the second highest cause of death in the world, despite the immense progress accomplished over the last 30 years by oncology researchers and specialists around the world.
Pierre Fabre has been a player in oncology since 1978 and has solid expertise in cancer of the lung, breast and bladder and more recently melanoma.
Today the Group makes oncology research its R&D priority.
Pierre Fabre brings its expertise in the research and development of molecules to the development of new biological entities and targeted biotherapies.
We place precision medicine at the heart of our strategy to provide the right treatment for the right patient at the right time by exploiting the synergies of molecular oncology and immunotherapy.
We take advantage of a global approach centered on the patient to strengthen our scientific understanding of the tumor microenvironment and our capacity to supply high-quality biological products based on multiple technological platforms such as monoclonal antibodies and immunoconjugates.
Dermatology and onco-dermatology R&D
The Pierre Fabre Group's dermatological research employs cross-disciplinary expertise in pharmaceutical and dermo-cosmetics research and development.
Dermatology R&D is the cornerstone which gives meaning to our continuum of activities "from health to beauty”.
The proven expertise of the Pierre Fabre Group in both oncology and dermatology makes onco-dermatology a natural and indispensable area of our R&D.
Today, our onco-dermatology research is centered on:
- Skin cancers: Melanoma and Basal cell carcinomas
- Skin lesions: Actinic keratosis
Consumer Health Care R&D
Consumer health care research at Pierre Fabre is above all based on the patient-consumer, to develop new products bringing greater well-being to homes around the world.
The consumer health care research conducted by the Pierre Fabre Group capitalizes on our pharmaceutical drug expertise and the galenic know-how of our dermo-cosmetics activities.
Innovation for family health is not restricted to the discovery of new formulas, it has the mission of bringing additional comfort to the patient-consumer. To achieve this, it works in close collaboration with marketing experts and favors short innovation cycles and the reuse of technologies.
Our ambition is therefore to remain at the forefront of the oral health (Elgydium, Inava, etc.), family health (Dexeryl range, Petit Drill, etc.) and natural health (Naturactive) sectors.
Focus on oncology: innovating to build a better future for patients
Today, the scientific expertise of the Pierre Fabre Group is renowned for its monoclonal antibodies and immunoconjugates.
This recognition places the company in a position to participate in the targeted biotherapies revolution.
Our ambition is to prioritize the development of new biological entities and targeted biotherapies which play a key role in tumor immunosuppression.
The research work conducted by our experts covers fields in which there are major medical expectations: skin, colon, breast and lung cancers.
ongoing clinical and discovery programs
partenariats signés avec des entreprises de biotechnologie depuis 2023

share of R&D spending devoted to oncology in 2025
A patient-centric approach: precision medicine
Today we tend to talk less about cancer and more about the mutations or abnormalities which characterize a cancer.
For example a mutation of the BRAF gene in colon cancer or melanoma will change both the prognosis of the patient and their treatment.
Targeted therapy:
In 2018, following the signature of a partnership with the American laboratory Array BioPharma, Pierre Fabre registered a new innovative targeted therapy combination in Europe for the treatment of melanoma.
A new targeted therapy combination is in development for colorectal cancer with a BRAF gene mutation.
Immunotherapy:
At Pierre Fabre, we think that the challenge of immuno-oncology is to provide the greatest possible benefit to the largest number of patients through greater exploitation of the power of the immune system at several levels.
To achieve this, we rely on a comprehensive approach centered on the cancer patient and supply high-quality biological products based on multiple technological platforms.
Two examples of innovations based on technological platforms.
- Example 1: Antibody + Cytotoxic molecule
An original molecule composed of an antibody specifically targeting certain tumors which overexpress a receptor that is important in terms of cancer cell proliferation (IGF1R), combined with a cytotoxic molecule, is currently undergoing trials in humans.
- Example 2: Checkpoint inhibitor
A treatment in the immune checkpoint inhibitor family is currently in the final phase of testing before it can be administered to cancer patients.
Building and being part of an ecosystem: partnerships
You go faster by yourself, you go further with others. At Pierre Fabre, we make this saying the guiding principle of our pharmaceutical R&D. Our history of innovation has been written in the plural since the creation of the Group almost 60 years ago.
Bringing new prescription drugs to patients, especially in the fight against cancer, requires increasingly lengthy, increasingly costly, research efforts. For the benefit of patients and health care professionals, we prefer to share knowledge and resources.
Our Inside-Out (working with external partnerships) and our Outside-In (accelerating the execution our internal projects) approach to partnership allows new therapeutic solutions to arrive on the market more quickly.
Want to initiate a partnership?
Join the R&D teams
Are you motivated by the challenge of research? You will find many offers on our website